
The microbiome undergoes marked changes during pregnancy; an active biological response to facilitate the metabolic and immunological adaptations required during pregnancy. Recent research has shown that the maternal microbiome has a significant influence on the baby’s microbiome in utero (Figure 1),5 with the transfer of organisms affecting a wide range of organ systems beyond the gastrointestinal tract (GIT).
The foetus is first exposed to commensal bacteria from the maternal gut/bloodstream that crosses the placenta and enters the amniotic fluid.6 Colonisation continues at birth with an infant’s GIT tending towards a skin-like (caesarean section) or a vaginal-like (vaginal delivery) configuration. Breastfeeding then provides a continuous supply of commensal bacteria to the infant,7 with the introduction of solid foods further increasing gut microbiota diversity.
Strain Specific Probiotics for Pregnancy, Breastfeeding and Infant Health combines Lactobacillus rhamnosus (LGG®), Bifidobacterium animalis ssp lactis (BB-12®), Bifidobacterium breve (M-16v) and Bifidobacterium longum (BB536), which have been shown to influence both the maternal and infant gastrointestinal microbiota. Furthermore, these strains support both immune and neurological development in the infant, representing an essential intervention for primary disease prevention.
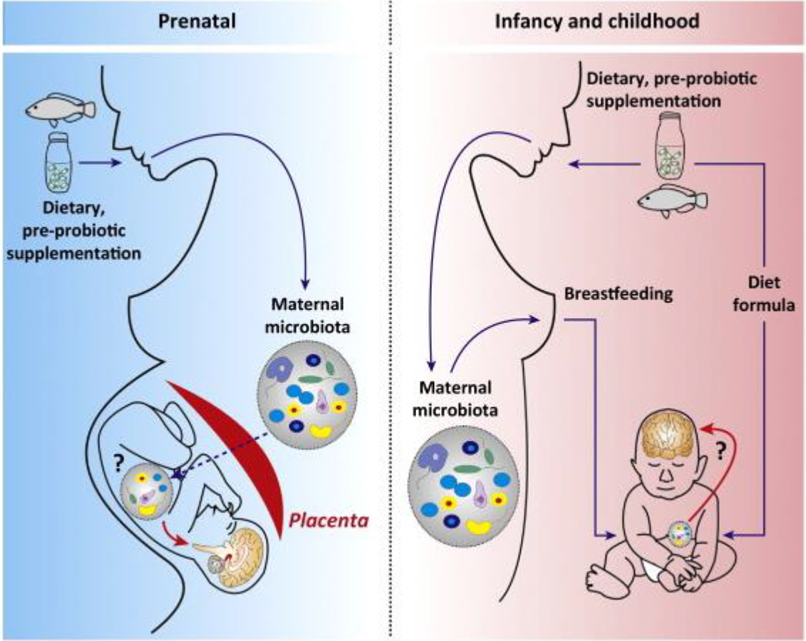
Figure 1: Transfer of the maternal microbiota to the infant and related factors.8
Creating Intelligent Immunity
The presence of a healthy and diverse gut microbiota from birth is paramount in establishing an appropriate immune system response throughout life. The probiotic strain, BB536 has been shown to help establish a healthy intestinal microbiota and improve T helper 1 (Th1) immune responses through enhancement of interferon gamma (IFN-γ) secretion.9 Similarly, oral administration of the strain M-16v has demonstrated suppression of T helper 2 (Th2) immune responses and IgE production, along with modulating systemic Th1/Th2 balance.10
Further to this, supplementation with LGG® pre- and post-natally enhances the immune benefits of breastfeeding. In one study, 20 billion CFU/day of LGG® was shown to prevent atopic eczema in infants when administered to the mother before delivery and during breastfeeding, with the preventive effects extending up to seven years of age.11 Similarly, BB-12® (in combination with LGG®) has demonstrated immunomodulatory actions via an increase in transforming growth factor-beta 2 (TGF-β2) in breast milk.12 TGF-β2 has been shown to reduce allergic inflammation in the infant and is a key factor for increasing secretory immunoglobulin A (sIgA) production in the mucosa, thereby helping to induce oral tolerance.13 This is significant as atopic mothers have lower concentrations of TGF-β2 in their breast milk.14
Moreover, the combination of LGG® and BB-12® has been shown to reduce the risk of respiratory tract infections in infants. In a randomised, double-blind, placebo-controlled study, newborns requiring formula before two months of age were given formula supplemented with LGG® and BB-12® (10 billion CFU/day of each strain) or placebo. Significant reductions were observed in acute otitis media, recurrent respiratory infections and antibiotic use,15 suggesting probiotics may offer a safe means of reducing the risk of respiratory tract infections during the first year of life.
Bacteria For Better Behaviour?
Similarly bacterial exposure and colonisation patterns are crucial for nervous system development. Following bacterial colonisation, increased production of serotonin (5-HT) and γ-aminobutyric acid (GABA), has been observed in the gut.16 These changes are integral to gut homeostasis and to programming of the hypothalamic–pituitary–adrenal (HPA) axis,17 which plays an important role in a healthy stress response.
In an ongoing, randomised, double-blind, placebo-controlled, prospective follow-up study of 159 mother-infant pairs, LGG® (10 billion CFU/day) was given to pregnant mothers four weeks before expected delivery and for six months after delivery (if not breastfeeding, LGG® was given directly to the infant). At 13 years of age, ADHD or Asperger’s Syndrome (AS) was diagnosed in 6/35 (17.1%) of children in the placebo group, with no children diagnosed in the probiotic group,18 a statistically significant difference. This trial highlights the potential for tailored probiotic supplementation to reduce the risk of certain behavioural complaints in children as they develop.
Don’t Forget About Mum
As mentioned previously, pregnancy is accompanied by metabolically adaptive processes that both maintain the pregnancy and help the baby grow and develop. Exaggerations in this adaptive process can lead some women to develop gestational diabetes, with associated clinical complications for both mother and foetus. In a randomised, double-blind, placebo-controlled trial, 256 normoglycaemic pregnant women in their first trimester received either nutritional counselling with probiotics, nutritional counselling and placebo, or no placebo or nutritional counselling. The probiotic group received LGG® and BB-12® (10 billion CFU/day of each strain). Blood glucose concentrations were lowest in the nutritional counselling plus probiotic group, both during pregnancy and in the 12 months postpartum period,19 demonstrating the maternal metabolic benefits of probiotic supplementation in pregnancy.
Support For Mum And Bub
Strain Specific Probiotics for Pregnancy, Breastfeeding and Infant Health combines strains LGG®, BB-12®, M-16v and BB536 to provide probiotic support during the maternal and infant development period, influencing preferential outcomes both for the mother and her child. These effects are not limited to the gastrointestinal environment, but also encompass systemic immune responsiveness, neurological implications and metabolic homeostasis. As a healthy and diverse microbiota is important for long-term health outcomes, probiotic supplementation, beginning with the mother, is one of the easiest and safest strategies for primary disease prevention in the infant.
References
- Bosch AA, Levin E, van Houten MA, Hasrat R, Kalkman G, Biesbroek G, et al. Development of upper respiratory tract microbiota in infancy is affected by mode of delivery. EBioMedicine. 2016 Jul;31(9):336-45.
- Peters SA, Yang L, Guo Y, Chen Y, Bian Z, Du J, et al. Breastfeeding and the risk of maternal cardiovascular disease: a prospective study of 300 000 Chinese women. J Am Heart Assoc. 2017 Jun 1;6(6):e006081.
- Gomez-Gallego C, Garcia-Mantrana I, Salminen S, Collado MC. The human milk microbiome and factors influencing its composition and activity. Semin Fetal Neonatal Med 2016 Dec; 21(6):400-405.
- Nishimura RY, Barbieiri P, de Castro GS, Jordão AA, Perdoná GD, Sartorelli DS. Dietary polyunsaturated fatty acid intake during late pregnancy affects fatty acid composition of mature breast milk. Nutrition. 2014 Jun 30;30(6):685-9.
- Borre YE, O’Keeffe GW, Clarke G, Stanton C, Dinan T, Cryan J. Microbiota and neurodevelopmental windows: implications for brain disorders. Trends Mol Med. 2014 Sep;20(9):509-18.
- Walker WA. The importance of appropriate initial bacterial colonization of the intestine in newborn, child, and adult health. Pediatric Research. 2017 May;82(3):387-395.
- Fernández L, Langa S, Martín V, Maldonado A, Jiménez E, Martín R, et al. The human milk microbiota: origin and potential roles in health and disease. Pharmacological Research. 2013 Mar 31;69(1):1-10.
- Borre YE, O’Keeffe GW, Clarke G, Stanton C, Dinan T, Cryan J. Microbiota and neurodevelopmental windows: implications for brain disorders. Trends Mol Med. 2014 Sep;20(9):509-18.
- Wu BB, Yang Y, Xu X, Wang WP. Effects of Bifidobacterium supplementation on intestinal microbiota composition and the immune response in healthy infants. World J Pediatr. 2016 May;12(2):177-82.
- Inoue Y, Iwabuchi N, Xiao JZ, Yaeshima T, Iwatsuki K. Suppressive effects of bifidobacterium breve strain M-16V on T-helper type 2 immune responses in a murine model. Biol Pharm Bull. 2009 Apr;32(4):760-3.
- Kalliomäki M, Salminen S, Poussa T, Iosolauri E. Probiotics during the first 7 years of life: a cumulative risk reduction of eczema in a randomized, placebo-controlled trial. Allergy Clin Immunol. 2007 Apr;119(4):1019-21.
- Rautava S, Arvilommi H, Isolauri E. Specific probiotics in enhancing maturation of IgA responses in formula-fed infants. Pediatr Res. 2006 Aug;60(2):221-4.
- Huurre A, Laitinen K, Rautava S, Korkeamaki M, Isolauri E. Impact of maternal atopy and probiotic supplementation during pregnancy on infant sensitization: a double-blind placebo-controlled study. Clin Exp Allergy. 2008 Aug;38(8):1342-8.
- Huurre A, Laitinen K, Rautava S, Korkeamaki M, Isolauri E. Impact of maternal atopy and probiotic supplementation during pregnancy on infant sensitization: a double-blind placebo-controlled study. Clin Exp Allergy. 2008 Aug;38(8):1342-8.
- Rautava S, Salminen S, Isolauri E. Specific probiotics in reducing the risk of acute infections in infancy-a randomised, double-blind, placebo-controlled study. Br J Nutr. 2009 Jun;101(11):1722-6.
- Ghaisas S, Maher J, Kanthasamy A. Gut microbiome in health and disease: Linking the microbiome-gut-brain axis and environmental factors in the pathogenesis of systemic and neurodegenerative diseases. Pharmacol Ther. 2016 Feb;158:52-62.
- Ghaisas S, Maher J, Kanthasamy A. Gut microbiome in health and disease: Linking the microbiome-gut-brain axis and environmental factors in the pathogenesis of systemic and neurodegenerative diseases. Pharmacol Ther. 2016 Feb;158:52-62.
- Pärtty A, Kalliomäki M, Wacklin P, Salminen S, Isolauri E. A possible link between early probiotic intervention and the risk of neuropsychiatric disorders later in childhood: a randomized trial. Pediatr Res. 2015 Jun;77(6):823-8.
- Laitinen K, Poussa T, Isolauri E. Nutrition, Allergy, Mucosal Immunology and Intestinal Microbiota Group. Probiotics and dietary counselling contribute to glucose regulation during and after pregnancy: a randomised controlled trial. Br J Nutr. 2009 Jun;101(11):1679-87.

Recent Comments